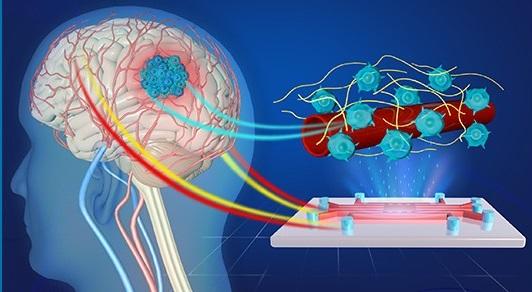
Creating ‘humanised’ in vitro models to study brain cancers using patient-derived cancer cells, could improve our understanding of how cancer sub-types and the microvessel environment drive chemo-resistance, leading to the rapid testing of new drugs and therapeutic approaches.
The current standard of care for patients diagnosed with glioblastoma – of which there are around 2,500 new cases every year in the UK alone – involves a combination of tumour resection surgery, chemotherapy and radiation that would give the patient an average survival rate of less than 15 months.
Brain tumours are known to be highly heterogeneous among different patients; and even for the same patient, the tumour composition can be very different depending on the tumour region, which means that different sub-types of cancer cells and their environment can respond very differently to the same drug.
In order to further understand the behaviour of patient-derived glioblastoma stem-like cells (GSCs) and their interactions with the blood-brain barrier – a key role in identifying new drug targets – there is a need to develop a microfluidic device to perform comparative studies in order to investigate how the types of GSCs and microvessels affect the cells’ gene expression, cancer cell speed and direction of migration. These GSCs can migrate in the brain vasculature within or away from the tumour mass, increasing tumour resistance to treatments and contributing to relapses. The results of the study, involving the University of Cambridge, University of Toronto, Radboud Institute for Molecular Life Sciences, King's College London and the University of Edinburgh, are published with open access in the journal Lab on a Chip.
The researchers created flowable vasculature models generated from endothelial cells from the human brain, umbilical vein and lung, in order to study the characteristics of the microvessels and their interaction with GSCs derived from different patient cancer subtypes. The customisable nature of the device, opens up the possibility for clinicians to be able to study tumour susceptibility to new treatments.
At Cambridge, this work was initiated between bioengineers Magda Gerigk, a recipient of the CRUK Multidisciplinary Studentship, and Dr Shery Huang, from the Department of Engineering; neurosurgeon Dr Harry Bulstrode, from the Department of Clinical Neuroscience; and clinician lead Professor David Rowitch, from the Department of Paediatrics.
Corresponding author Dr Huang, Assistant Professor in Bioengineering and Group Leader of the Biointerface Research Group, said the novel creation of the flowable brain microvessel, the ability to perform gene expression analysis, and the use of live-cell imaging and image analysis aided understanding of brain tumour progression.
"We look forward to seeing engineering tool kits making an increasing impact on clinical treatments," said Dr Huang.
Reference:
Magda Gerigk et al. ‘On-chip perivascular niche supporting stemness of patient-derived glioma cells in a serum-free, flowable culture’. Lab on a Chip (2021). DOI: 10.1039/D1LC00271F
Image: Artist's impression of the 3D microvessel-on-a-chip device for the study of glioblastoma.
Credit: Yuanyuan Bei
Reproduced courtesy of University of Cambridge, Department of Engineering