The brain is arguably the most important organ in the body, as it controls most other body systems and enables reasoning, intelligence, and emotion. Humans have evolved a variety of protective measures to prevent physical damage to the brain: it sits in a solid, bony case – the skull – and is wrapped in three layers of watertight tissue known as the meninges.
What has been less clear is how the body defends the brain from infection. Elsewhere in the body, if bacteria or viruses enter the bloodstream, our immune system kicks in, with immune cells and antibodies that target and eliminate the invader. However, the meninges form an impermeable barrier preventing these immune cells from entering the brain.
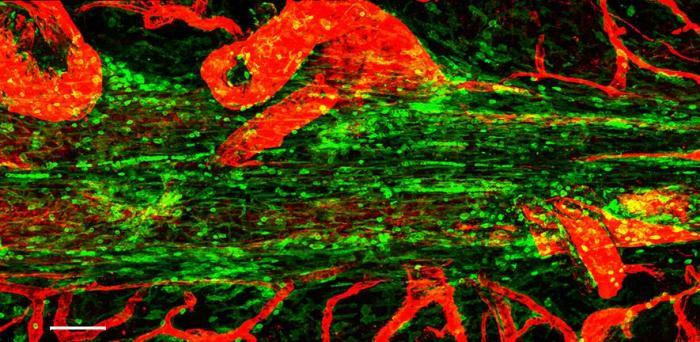
In research published in Nature, a team led by scientists at the University of Cambridge, UK, and the National Institute of Health, USA, have found that the meninges are home to immune cells known as plasma cells, which secrete antibodies. These cells are specifically positioned next to large blood vessels running within the meninges allowing them to secrete their antibody ‘guards’ to defend the perimeter of the brain. When the researchers looked at the specific type of antibody produced by these cells, they got a surprise – the antibody they observed is normally the type found in the intestine.
Plasma cells are derived from a particular type of immune cell known as a B cell. Every B cell has an antibody on its surface that is unique to that cell. If an antigen (the part of a bacterium or virus that triggers an immune response) binds to that surface antibody, the B cell becomes activated: it will divide to make new offspring that also recognise that same antigen.
During division, the B cell introduces a mutation into the antibody gene so that one amino acid is changed and its binding characteristics are slightly different. Some of these B cells will now produce antibodies that enable better binding to the pathogen – these go on to expand and multiply; B cells whose antibodies are less good at binding die off. This helps ensure the body produces the best antibodies for targeting and destroying particular antigens.
Normally, the antibodies found in the blood are a type known as Immunoglobulin G (IgG), which are produced in the spleen and bone marrow – these antibodies protect the inside of the body. However, the antibodies found in the meninges were Immunoglobulin A (IgA), which are usually made in the gut lining or in the lining of the nose or lungs – these protect mucosal surfaces, the surfaces that interface with the outside environment.
The team were able to sequence the antibody genes in B cells and plasma cells in the gut and meninges and show that they were related. In other words, the cells that end up in the meninges are those that have been selectively expanded in the gut, where they have recognised particular pathogens.
“The exact way in which the brain protects itself from infection, beyond the physical barrier of the meninges, has been something of a mystery, but to find that an important line of defence starts in the gut was quite a surprise,” said lead scientist Professor Menna Clatworthy from the Department of Medicine and CITIID at the University of Cambridge and the Wellcome Sanger Institute.
Image: Confocal micrograph showing the superior saggital sinus in the mouse. Immune cells are shown in green lining this tube, and blood vessels in red
Credit: Zach Fitzpatrick
Reproduced courtesy of the University of Cambridge